Quick outline
- Why wholesale lumbrokinase sourcing is getting more attention
- What lumbrokinase actually is in a B2B buying context
- The supplier questions that matter before price talks
- How to read specs, activity, and processing details without getting lost
- Common risks in wholesale supply—and how smart buyers reduce them
- What a reliable supplier relationship usually looks like
- FAQs for bulk buyers

If you’re sourcing lumbrokinase at wholesale level, you already know this isn’t a simple “compare price per kilo and move on” kind of ingredient. Not even close.
Lumbrokinase sits in that interesting space where science, sourcing, manufacturing, and compliance all collide. For supplement brands, nutraceutical formulators, pharmaceutical ingredient buyers, and health product wholesalers, it can look like a promising high-value ingredient. But the buying process? That’s where things get real. One supplier may talk about purity, another may emphasize activity units, and a third may offer a very attractive quote that somehow leaves out the details you actually need.
Here’s the thing: with lumbrokinase, the details are the product.
A bulk buyer needs more than a sales sheet. You need to know what the raw material is, how it’s produced, how activity is verified, how stable it is during storage and formulation, and whether the documentation can hold up when your QA team starts asking hard questions. That matters because lumbrokinase is widely discussed for its fibrinolytic and thrombolytic properties in research and review literature, and the way it is extracted and processed directly affects how buyers evaluate performance and safety.
So let me explain what buyers really need to look at before signing a wholesale supply deal.
Why more buyers are paying attention
Lumbrokinase is a group of fibrinolytic enzymes derived from earthworms and has been described in the literature as a potent bioactive component with antithrombotic and thrombolytic relevance. Review literature also notes interest in oral formulations and clinical translation in some markets, which is one reason ingredient buyers keep it on their radar.
That growing attention makes sense. Buyers in this category are not usually looking for mass-market commodity proteins. They’re looking for ingredients that can support differentiated product positioning. Lumbrokinase often enters the conversation when brands want a more specialized cardiovascular-support or circulation-focused ingredient story.
But let’s be honest—specialized ingredients also attract uneven supply quality.
That’s why wholesale sourcing has to start with one simple question: what exactly is the supplier selling you?
First, know what you’re buying
This sounds obvious, but it’s where many sourcing problems begin.
Some sellers use “earthworm protein,” “earthworm peptide,” “earthworm extract,” and “lumbrokinase” almost as if they’re interchangeable. They’re not. Not at all.
Earthworm-derived materials can include broad protein powders, peptide fractions, extracts, and more refined enzyme-focused ingredients. The literature around earthworm proteins and peptides covers a wide range of bioactivities—antioxidant, ACE inhibitory, immunomodulatory, antimicrobial, and fibrinolytic—so a buyer has to separate general earthworm-derived raw materials from lumbrokinase-specific enzyme preparations.
In plain English: not every earthworm-based ingredient is lumbrokinase, and not every earthworm powder quote is relevant if your target is enzyme activity.
That distinction affects:
- positioning
- potency expectations
- testing methods
- regulatory strategy
- formulation behavior
- cost structure
And yes, it affects whether your procurement team is comparing apples to apples or apples to, well, mud.
Don’t start with price—start with the spec sheet
A wholesale buyer should always ask for the specification sheet before discussing volume discounts in any serious way. Price matters, of course. But if one supplier’s material has lower verified activity, looser microbial limits, or weaker traceability, the cheaper quote may not actually be cheaper once you calculate usable value.
When reviewing a lumbrokinase spec, pay close attention to these points.
Activity units are not background noise
For lumbrokinase, activity is central. Buyers are not just purchasing weight. They are purchasing enzymatic function.
That means you need clarity on:
- the activity unit used
- the assay method used to determine that activity
- whether the stated activity is minimum, typical, or release value
- whether the result is tested per gram, per batch, or under a specific formulation condition
If a supplier cannot explain how activity is measured, that’s not a small gap. That’s a major warning sign.
Research and industry materials consistently frame lumbrokinase as a fibrinolytic enzyme system, so the relevance of activity-based testing is obvious. More broadly, supplier materials around earthworm-derived ingredients also emphasize functional performance rather than only crude composition.
Honestly, this is where experienced buyers separate themselves from first-time buyers. They don’t just ask, “What’s the potency?” They ask, “How was that potency established?”
Raw material origin matters more than many buyers expect
The source organism, breeding conditions, and upstream handling can all influence consistency. Some supplier-side production information for earthworm protein identifies the raw material source and basic processing flow, including selection, separation, washing, filtration, low-temperature drying, sterilization, and packaging.
That kind of production transparency matters because it helps answer bigger questions:
Was the raw material farmed or wild collected?
Was species identity controlled?
Was the process designed for food, nutraceutical, or pharmaceutical expectations?
How is contamination risk managed during cleaning and separation?
A buyer doesn’t need a dramatic factory tour story. You need documented control points.
And if the supplier gets vague right when you ask about origin, that usually tells you something.
The process is not just a technical footnote
For enzyme ingredients, production method shapes the final material. Extraction, separation, drying conditions, purification approach, and stabilization strategy can all influence performance.
Even in broader earthworm-protein research, processing methods like digestion, fractionation, filtration, and chromatography are critical to isolating bioactive fractions and identifying active peptides.
That does not mean every wholesale buyer needs a doctoral-level process map. But you do need enough detail to judge whether the supplier has a controlled, repeatable manufacturing system rather than a loose extraction story wrapped in marketing language.
Ask these questions:
- Is the enzyme fraction standardized?
- Is the product spray-dried, freeze-dried, or otherwise stabilized?
- What excipients or carriers are used?
- Is maltodextrin, starch, or another support material included?
- What is the true active-content basis versus finished powder basis?
That last point is easy to miss. A powder may contain an active enzyme fraction plus carriers. That is not automatically bad. It just needs to be disclosed clearly.
Documentation is part of the product
For wholesale supply, documents are not paperwork after the fact. They are part of the material offering itself.
At minimum, serious buyers usually want to review:
- COA
- specification sheet
- microbiological testing
- heavy metal testing
- allergen statement
- residual solvent statement, where relevant
- non-GMO statement, where relevant
- TDS or product data sheet
- manufacturing flow summary
- storage and shelf-life guidance
If the supplier serves export markets, you may also need batch traceability documents, country-of-origin documentation, and market-specific declarations.
This is especially important because earthworm-based ingredients sit in a category where regulatory treatment can vary by market and application. The scientific literature describes earthworm proteins as food-resource candidates in some contexts and medicinally relevant extracts in others, which tells buyers right away that regulatory positioning is not one-size-fits-all.
In other words, your supplier doesn’t just need product knowledge. They need document discipline.

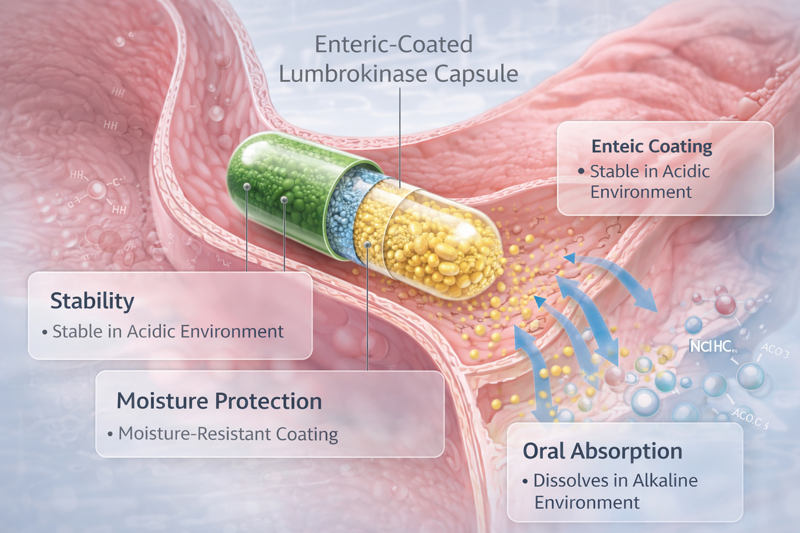
Quality risk is often hiding in the “good enough” supplier
This is the uncomfortable part. Many wholesale supply problems don’t come from obviously bad suppliers. They come from suppliers that are almost good enough.
The sample looks okay.
The price is competitive.
The email replies are fast.
The assay looks plausible.
But when scale-up starts, the cracks show—batch variability, missing test details, inconsistent odor or color, unstable activity, or delayed documentation.
You know what? That’s why pilot verification matters.
Before placing a large order, buyers should run a controlled qualification process:
- Review documents
- Evaluate sample performance
- Check batch consistency where possible
- Confirm packaging and storage conditions
- Verify commercial terms and lead time
Not glamorous, but very necessary.
MOQ, lead time, and packaging aren’t boring details
Procurement teams sometimes focus so hard on active specs that they underweight operational fit. Then the trouble starts later.
Ask about:
- minimum order quantity
- standard lead time
- stock versus made-to-order supply
- packaging size options
- inner and outer packaging materials
- cold-chain or temperature-sensitive shipping needs
- shelf life under unopened conditions
- retest period, if applicable
For specialty enzyme ingredients, poor packaging or unclear storage guidance can quietly damage value before the material ever reaches production.
That’s frustrating because it’s preventable.
A trustworthy supplier should be able to talk to QA, not only sales
This one is big.
A real supplier should be able to support technical discussions with your quality team, not only commercial chats with your purchasing manager. If your questions about assay method, batch release criteria, or production controls keep getting answered with broad sales phrases, you probably don’t have enough technical support behind the account.
Strong suppliers usually show their strength in the unflashy moments. They answer slowly maybe—but clearly. They explain limits. They distinguish between tested data and estimated data. They tell you what they can support and what they can’t.
That’s not weak selling. That’s mature supply behavior.
Regulatory fit depends on your target market
This is where many buyers need to pause and think strategically.
A lumbrokinase ingredient that works for one channel or region may not be suitable for another without extra review. Product category, dosage form, label language, claims, import rules, and ingredient classification can all change the path.
The research base around earthworm-derived bioactives is broad, covering food applications, bioactive peptides, and pharmacological effects, but research interest is not the same thing as automatic regulatory clearance in every market.
So buyers should ask early:
- What applications does the supplier commonly support?
- Is the material positioned as food ingredient, supplement ingredient, or another category?
- What export documents are available?
- What claims should be avoided?
- Has the supplier shipped to your destination market before?
A good supplier won’t pretend every market works the same way.
What smart buyers usually ask before committing
By the time you’re close to placing an order, your question list should be pretty sharp. It often includes things like:
“Can you provide the exact activity test method and release standard?”
“What carriers are included in the final bulk powder?”
“How do you control batch-to-batch variation?”
“What is the shelf life under recommended storage?”
“Can you provide recent COAs from commercial batches?”
“What is your standard MOQ and production lead time?”
“What packaging format do you use for export shipments?”
“Can your QA team support our technical review?”
Simple questions, but revealing ones.
The answers tell you whether the supplier is really operating at ingredient level—or just hoping you won’t look too closely.
Wholesale sourcing is really about consistency
That’s the core lesson.
Lumbrokinase is not the kind of ingredient where one good sample solves everything. Bulk buyers need repeatability. Same activity range. Same documentation standard. Same basic sensory profile. Same handling performance. Same lead-time reliability.
Because once the ingredient moves into your own production system, inconsistency becomes your problem.
And that gets expensive fast.
So, where should buyers focus first?
If you’re evaluating lumbrokinase wholesale supply, focus on four priorities first:
1. Verified enzyme activity
Without clear, defensible activity data, the rest of the offer is shaky.
2. Manufacturing transparency
You need a basic but credible understanding of source material and processing flow. Supplier-side earthworm protein manufacturing information shows how useful even a concise process summary can be for evaluating control points.
3. Documentation quality
A supplier who struggles with documents during qualification usually won’t get easier during commercial supply.
4. Commercial reliability
MOQ, lead time, packaging, and responsiveness matter. Specialty ingredients live or die on execution.
Final thought
Buying lumbrokinase in bulk is not just a sourcing task. It’s a filtering task.
You are filtering out vague specs, weak documentation, unclear activity claims, and suppliers who sound confident but can’t support technical review. The buyers who do this well are not necessarily the ones who buy cheapest. They’re the ones who buy cleanly—clean data, clean process, clean communication.
That’s what reduces risk. That’s what protects product quality. And that’s what makes a wholesale relationship actually work.
FAQs
1. What should buyers check first when sourcing lumbrokinase wholesale?
Start with the specification sheet, enzyme activity data, assay method, and batch documentation. For bulk lumbrokinase supply, activity verification is usually more important than price alone.
2. Is lumbrokinase the same as earthworm protein powder?
No. Lumbrokinase is a fibrinolytic enzyme complex derived from earthworms, while earthworm protein powder is a broader raw material category. Buyers should not assume an earthworm protein wholesale offer is equivalent to a lumbrokinase bulk ingredient.
3. Why are activity units important in lumbrokinase bulk purchasing?
Because lumbrokinase is bought for enzymatic function, not only for weight. A wholesale buyer needs to know how activity was measured, what standard was used, and whether the stated value reflects release testing.
4. What documents should a lumbrokinase supplier provide for B2B orders?
Most buyers ask for a COA, product specification, microbiological results, heavy metal results, storage guidance, shelf-life information, and batch traceability documents. Export buyers may also need origin and market-specific declarations.
5. How can buyers reduce risk when choosing a lumbrokinase wholesale supplier?
Use a sample qualification process, review recent batch documents, confirm packaging and storage conditions, and make sure the supplier can support technical questions from QA or regulatory teams. That’s usually the safest way to assess bulk lumbrokinase suppliers before scaling up.
