
Before anything else, here’s the honest answer: yes, but only if you stop treating it like one product for one world market.
That sounds a bit contradictory, I know. Lumbrokinase has real commercial appeal. It already carries a long East Asian use history, and China’s NMPA records show registered lumbrokinase capsule products, re-registration records, inspection results, and API/GMP references tied to lumbrokinase manufacturing. At the same time, the same ingredient can run into very different rules once it leaves that context and enters the U.S., EU, or other supplement-led markets.
So the question is not really, “Can lumbrokinase go global?”
It’s more like, “Which version of lumbrokinase can go where, under which legal identity, with which claims, and with what evidence package?”
That’s the real game. And for suppliers, manufacturers, and brand owners, getting that right early can save a lot of wasted formulation work later.
First, what makes lumbrokinase commercially interesting?
Lumbrokinase is generally described as a group of fibrinolytic enzymes derived from earthworm extracts, and that matters because buyers rarely look at it as “just another enzyme.” They look at it as a circulation-support, cardiovascular-positioned, or sometimes pharma-adjacent ingredient. That gives it market pull — but it also creates regulatory tension fast, because the more the story leans toward thrombosis, clot breakdown, stroke, DVT, or post-ischemic care, the closer it gets to drug territory in many markets. FDA materials also show the agency has been studying lumbrokinase in its toxicology and regulatory science work, which is a reminder that the ingredient sits in a higher-scrutiny bucket than ordinary commodity supplements. (U.S. Food and Drug Administration)
That’s why lumbrokinase often feels like a promising ingredient wrapped in legal fine print. Exciting, yes. Frictionless, no.
Regional fit at a glance
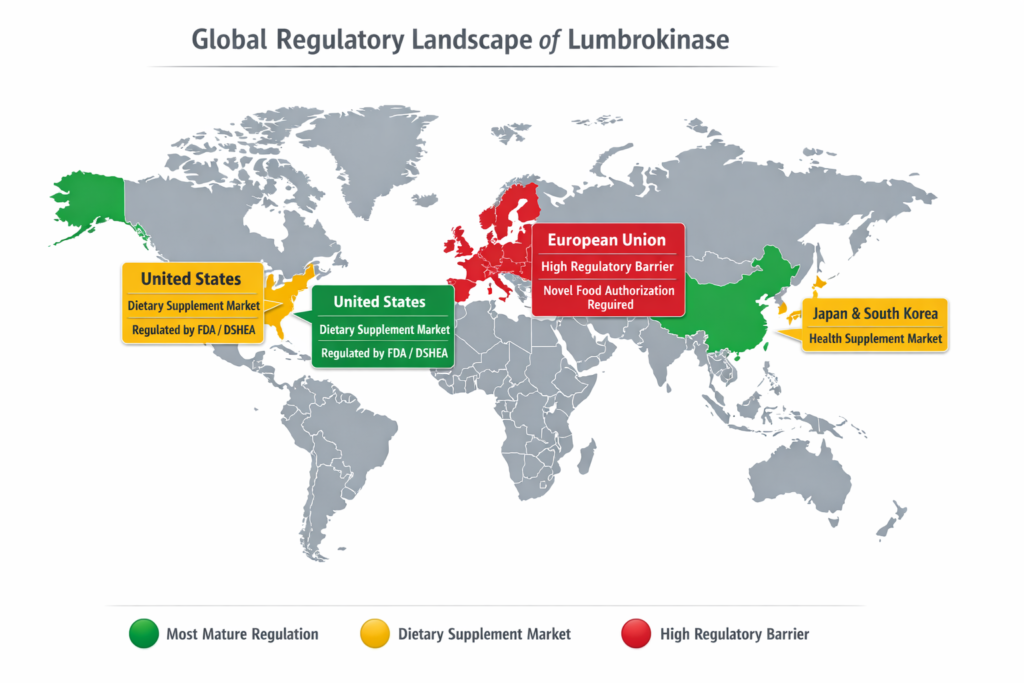
If you’re looking at broad commercial suitability, the markets do not line up evenly.
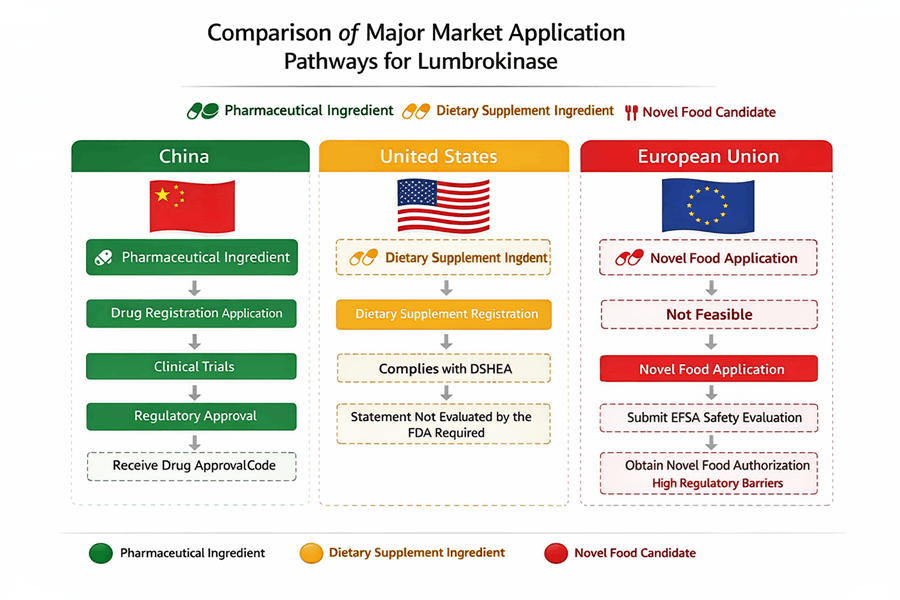
China looks like the most natural fit because lumbrokinase already appears in official drug-related records, including registered products and manufacturing oversight trails.
The United States can be workable, especially for a dietary supplement route, but only with disciplined labeling, structure/function claims, manufacturing controls, and a careful ingredient-history review. FDA makes clear that disease claims and treatment claims are a different animal altogether. (U.S. Food and Drug Administration)
The European Union is the toughest mainstream target for food or supplement positioning, because novel food rules can apply where there is no demonstrated significant pre-1997 consumption, and the Commission also notes that certain substances may fall instead under food enzyme, additive, flavoring, or medicinal-product frameworks depending on intended use. (Food Safety)
So yes, lumbrokinase is suitable for different regional markets — but not equally, and not under one universal sales script.
United States: the market is open, but the claim box is tight
Here’s the thing. In the U.S., the biggest commercial opportunity is usually not a drug-style pitch. It’s a dietary supplement positioning with tightly controlled claims.
FDA distinguishes structure/function claims from disease claims. A statement about supporting normal circulation or helping maintain healthy cardiovascular function lands very differently from a statement about dissolving clots, reducing thrombosis, treating stroke risk, or managing DVT. The first can be viable in supplement language if truthful and properly handled; the second can move the product toward unapproved drug territory. FDA also requires notification for certain structure/function claims within 30 days after first marketing, and supplement manufacturers are subject to 21 CFR Part 111 cGMP rules. (U.S. Food and Drug Administration)
There’s another layer buyers shouldn’t ignore: ingredient status. FDA explains that any dietary ingredient not marketed in a U.S. dietary supplement before October 15, 1994 may be a new dietary ingredient, and manufacturers/distributors must evaluate whether an NDI notification is needed. FDA also says there is no authoritative master list of ingredients marketed before that date. That uncertainty is exactly why lumbrokinase launches need legal review before the first commercial PO, not after. (U.S. Food and Drug Administration)
And then there’s safety surveillance. If a supplement is sold in the U.S., adverse-event reporting obligations matter, especially for a cardio-positioned enzyme that may be used by consumers already taking anticoagulants or antiplatelet drugs. (U.S. Food and Drug Administration)
So, is lumbrokinase suitable for the U.S.?
Yes, potentially — as a carefully managed supplement ingredient, not as a freewheeling therapeutic claim engine.
European Union: likely the hardest market
Honestly, the EU is where a lot of ingredient plans go from confident to cautious.
The European Commission’s Novel Food status Catalogue says the catalogue is a non-binding orientation tool, but it also states that a business placing a product on the market must be able to prove a history of significant consumption before 15 May 1997 if it wants to avoid the novel food route. The Commission also notes that depending on intended use and technological function, a substance might instead fall under rules for food additives, flavorings, food enzymes, or medicinal products. (Food Safety)
That creates two headaches for lumbrokinase sellers.
The first is history of consumption. Earthworm-derived ingredients are not the sort of everyday EU legacy ingredient that glides through on assumption alone. If you can’t prove the right history for the specific form, processing method, and intended use, you may be staring at a novel-food strategy. (Food Safety)
The second is medicinal positioning risk. Lumbrokinase is commercially attractive precisely because buyers associate it with fibrinolytic and antithrombotic language. But that same language can become a problem. Once marketing starts sounding therapeutic, the food/supplement pathway gets shaky.
That doesn’t mean the EU is impossible. It means it is usually documentation-heavy, slower, and less forgiving than Asia or the U.S. for this ingredient.
For many B2B companies, the practical answer is to treat the EU as a second-phase market, not the first export market.
China: the most natural home market
If lumbrokinase has a home-field advantage anywhere, it is in China.
NMPA search results show multiple official references to lumbrokinase API and finished dosage forms, including enteric capsules and tablets, GMP-related records, inspection data, and re-registration records. In plain English, this is not a concept ingredient in China. It has a visible regulatory and manufacturing footprint.
That doesn’t mean “easy.” It means legible.
And legible is valuable. Buyers like markets where an ingredient already has a known regulatory identity. It reduces the amount of education needed with distributors, importers, physicians, or channel partners. It also helps with dossier building, specification design, and supplier qualification.
For Chinese manufacturers thinking globally, this matters in another way too. China may be the strongest place to build manufacturing credibility first — standardized activity, impurity controls, raw-material traceability, microbial limits, and batch reproducibility — before adapting the commercial format for export markets.
In other words, China can function as both a sales market and a validation market.
Asia-Pacific beyond China: promising, but not automatic
This is where things get interesting. A lot of APAC markets are commercially receptive to enzyme ingredients, traditional-health stories, and practitioner-led channels. That sounds like good news, and it is — partly.
But “APAC” is not one rulebook. Some markets lean toward food supplement treatment, some push borderline products toward medicine registration, and some care a lot about traditional use while still demanding modern safety and quality files. So a lumbrokinase product may be commercially attractive in the region, yet still require country-by-country repositioning.
That’s why the best export strategy is often not one master label translated into five languages. It’s a regional base dossier plus local claim adaptation, local labeling review, and local channel-specific positioning.
A neat idea on paper can become a customs problem in real life. Happens all the time.
The real issue isn’t only regulation — it’s product identity
This part gets overlooked.
When buyers ask whether lumbrokinase suits different regional markets, they often focus only on the ingredient. But regulators usually look at the finished product identity:
- Is it a drug, supplement, food ingredient, or borderline product?
- Is the claim therapeutic, functional, or general wellness?
- Is the active standardized by enzyme activity units?
- Is the raw material clearly identified by species and process?
- Is the dosage form making the product look more pharmaceutical than nutritional?
- Are there interaction warnings, especially around anticoagulants?
See the pattern? The ingredient doesn’t travel alone. It travels with its story, spec sheet, claims, and risk profile.
That’s why two companies can sell “lumbrokinase” and get very different outcomes in different markets.
Claim strategy: where many launches go sideways
Let me explain the common trap.
A company builds a nice formula, gets excited about the science, and starts writing copy like “dissolves blood clots,” “prevents stroke,” or “works like a natural thrombolytic.” Commercially, that may sound punchy. Regulatory-wise, it’s asking for trouble in many jurisdictions.
In the U.S., FDA draws a clear line between structure/function claims and disease claims. In the EU, therapeutic-style positioning can intensify medicinal-borderline issues. In any market, bold cardio claims also raise safety questions about concurrent use with anticoagulants, antiplatelets, or perioperative settings. (U.S. Food and Drug Administration)
So the smarter route is usually boring in the best possible way:
support language, normal-function language, quality language, and professional-use context where appropriate.
Not flashy. But bankable.
What buyers should do before launch
If you’re sourcing or formulating lumbrokinase for multiple regions, don’t start with the brochure. Start with the regulatory build.
You need, at minimum, a clear species identity, extraction and purification description, activity standardization method, contaminant and microbiology controls, stability data, and a claim matrix by country. For the U.S., add an ingredient-history/NDI review and label-claim review. For the EU, add a novel-food and product-classification screen at the very beginning. For China, make sure your documentation lines up with the regulatory identity you’re actually using in-market. (U.S. Food and Drug Administration)
That may sound like a lot. It is. But it’s still cheaper than relabeling, reformulating, or having inventory stranded.
So, is lumbrokinase suitable for different regional markets?
Yes — selectively, strategically, and with market-specific positioning.
It looks strongest where there is already medical, traditional, or official familiarity with the ingredient, especially China. It looks commercially workable in the United States if handled as a disciplined dietary supplement ingredient with careful claims and compliance review. It looks far more demanding in the European Union, where novel-food and classification questions can raise the bar quickly.
So the better conclusion is not “global yes” or “global no.”
It’s this: lumbrokinase is suitable for different regional markets only when the regulatory pathway, evidence package, and claim language are tailored to each market from day one.
That’s not a limitation, really. It’s the business model.
FAQs
1. Can lumbrokinase be sold as a dietary supplement in the U.S.?
Potentially yes, but the product must stay within dietary-supplement rules, use compliant structure/function claims, follow supplement cGMP requirements, and undergo an ingredient-status review to assess whether an NDI notification may be needed. (U.S. Food and Drug Administration)
2. Is lumbrokinase easier to market in China than in Europe?
Generally, yes. China has official records showing lumbrokinase in registered product and manufacturing contexts, while the EU may require proof of significant pre-1997 consumption or a novel-food assessment, plus a product-classification review.
3. Why is claim wording so important for lumbrokinase products?
Because claims can change the product’s regulatory identity. Phrases about treating thrombosis, dissolving clots, or preventing stroke can move a lumbrokinase product toward drug territory in many jurisdictions. FDA explicitly separates structure/function claims from disease claims. (U.S. Food and Drug Administration)
4. Does the EU automatically ban lumbrokinase supplements?
No, not automatically. But the EU does require businesses to prove relevant consumption history or pursue the appropriate authorization path, and some intended uses may trigger other regulatory frameworks besides novel food. (Food Safety)
5. What is the best long-tail strategy for launching lumbrokinase in global markets?
The best long-tail strategy for launching lumbrokinase in global markets is to build one strong technical dossier, then localize regulatory classification, claims, labeling, and evidence expectations country by country rather than trying to sell one universal lumbrokinase product everywhere. (U.S. Food and Drug Administration)
