Brief outline
- Why buyers get tripped up by “purity”
- What lumbrokinase actually is, and why activity units matter more
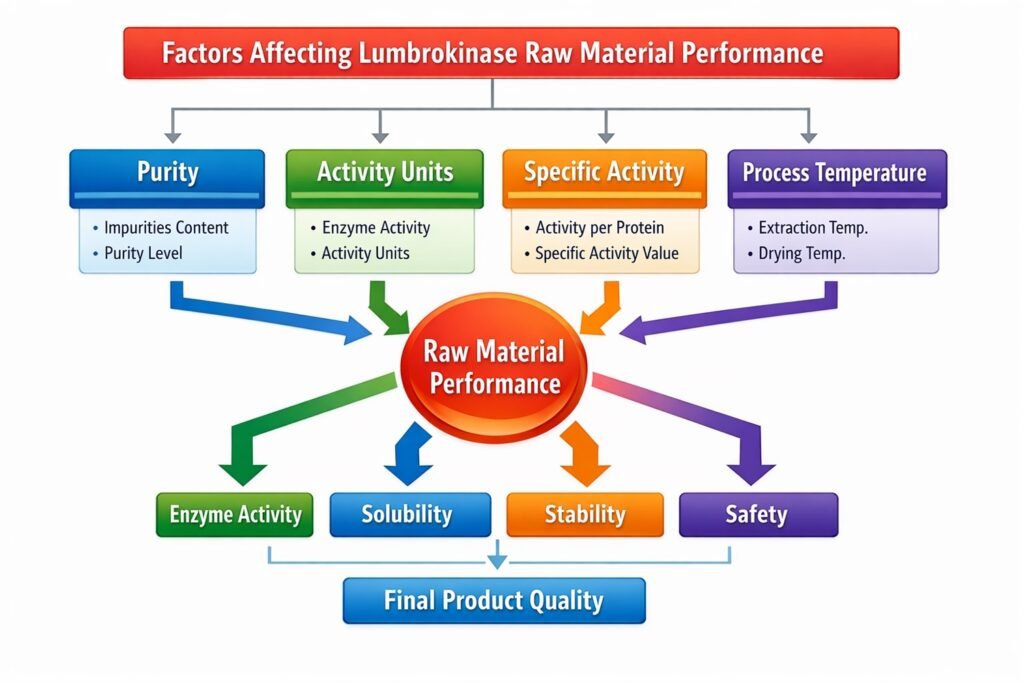
- Purity vs potency vs specific activity
- How lower-purity material can sometimes outperform a cleaner-looking powder
- What sourcing teams should ask suppliers
- Red flags in COAs and test methods
- Practical sourcing advice for supplements, nutraceuticals, pharma, and ingredient trade
- FAQ

When buyers source lumbrokinase, “purity” sounds like the obvious shortcut. Higher purity should mean better performance, right?
Well, yes—and no.
That little contradiction is where a lot of sourcing mistakes begin. In the real market, purity matters. Of course it does. But for lumbrokinase, purity alone does not tell you whether the ingredient will perform well in a finished product, hold up through handling, or deliver the fibrinolytic activity your customer expects. What matters more, often a lot more, is how purity connects to activity units, specific activity, and test method consistency.
Here’s the thing: buyers don’t purchase lumbrokinase because it looks clean on paper. They buy it because they need measurable enzymatic performance.
So let’s talk about what purity really means—and what it definitely does not mean.
A quick reality check: lumbrokinase is not “just one clean molecule”
Lumbrokinase is generally described as a group of fibrinolytic enzymes derived from earthworm extract, not a single uniform compound. A review in Animal Models and Experimental Medicine describes lumbrokinase as a fibrinolytic enzyme group first isolated from Lumbricus rubellus, with thrombolytic activity linked to fibrin specificity and a lower tendency toward hyperfibrinolysis-related bleeding compared with less selective systems.
That matters for sourcing.
Why? Because when buyers say “purity,” they may be talking about several different things at once:
- protein purity
- enzyme fraction enrichment
- removal of non-active proteins
- lower ash, moisture, or carrier load
- absence of fillers or denatured protein
Those are not the same thing. Not even close.
And if a supplier cannot explain which one they mean, you’re already in shaky territory.
Why specific activity deserves more attention than headline purity
The strongest clue in your materials is the testing document for lumbrokinase activity. It states that, on a dry basis, the product should have not less than 12,000 units per mg, and not less than 20,000 units of specific activity per mg protein. It also defines specific activity as:
specific activity = units per mg of sample / mg protein per mg of sample.
That’s the heart of the issue.
A high-purity ingredient that carries weak enzymatic performance is not a strong lumbrokinase ingredient. It’s just a cleaner-looking powder. Meanwhile, a material with well-controlled protein composition and high verified specific activity may outperform a “purer” material in actual fibrin plate testing.
So, when purity rises, performance only improves if the active fibrinolytic enzyme fraction is preserved and concentrated.
That last part is easy to miss. And honestly, plenty of buyers do miss it.
Purity helps performance—when it removes the right things
Let’s be fair. Purity is not meaningless. Better purification can improve performance in several practical ways.
First, it can reduce inactive protein ballast. If you remove non-functional protein and keep the active enzyme system intact, the final material can show higher activity per milligram and better batch consistency.
Second, it can lower interference in formulation. Cleaner material may behave better in capsules, tablets, and blends. It may also make the COA easier to interpret because there is less noise from unrelated solids.
Third, it can improve reproducibility. And for pharma-facing or export-facing buyers, reproducibility is gold. Not glamorous, but gold.
Still, that only works if purification does not strip out the wrong fractions or damage the enzyme.
That’s where some sourcing programs get burned.
When “more pure” can actually mean “less useful”
Enzymes are touchy. They care about heat, pH, moisture, and processing stress more than marketing copy ever will.
One of your uploaded files notes that lumbrokinase loses activity above 60°C, and that spray drying in earthworm protein or peptide production can deactivate the enzyme. That point is huge for buyers. A supplier can show a cleaner protein fraction after aggressive downstream processing, but if activity has been knocked down during drying or handling, then performance suffers where it counts.
So yes, a more refined ingredient can be worse if:
- purification is too harsh
- drying conditions denature the enzyme
- storage moisture is poorly controlled
- fillers are removed but active fractions are also lost
- the assay method overstates potency relative to practical use
That’s why experienced buyers ask a slightly annoying but very smart question:
“Higher purity according to which definition, and tied to which activity assay?”
If the supplier hesitates, that tells you something.
The assay method changes the purity conversation
Another point from the same activity document is just as important as the unit number itself: the activity is measured by a fibrin plate style method using fibrinogen, thrombin, agarose gel, standard curves, and lysis zone measurement after incubation.
That means lumbrokinase performance is not judged by generic protein content alone. It is judged by functional fibrinolytic effect.
And that’s a big distinction in commercial buying.
Because two suppliers may both claim “98% purity,” but if one verifies performance with a well-controlled fibrin-based assay and the other leans on a broad protein assay or vague internal standard, you are not comparing like with like. Not at all.
Honestly, this is where many price comparisons fall apart. They look neat in a spreadsheet, then messy in production.

What buyers should look at besides purity
If you source for dietary supplements, nutraceuticals, pharmaceutical intermediates, or contract manufacturing, keep purity in the file—but don’t let it run the meeting.
A more useful evaluation stack looks like this:
1. Activity units per mg
This tells you the performance density of the powder. The uploaded activity method sets a dry-basis threshold of not less than 12,000 units per mg.
2. Specific activity
This is even more revealing because it connects potency to actual protein mass, not just total powder weight. The same file sets not less than 20,000 units per mg protein as the specific activity floor.
3. Moisture and drying loss
The method document limits loss on drying to not more than 5.0%. That matters because excess moisture can distort apparent potency, harm stability, and create batch-to-batch drift.
4. Process temperature control
If upstream or downstream processing crosses the threshold where enzyme activity drops, the cleanest powder in the room may still be the weakest one. Your internal materials state that lumbrokinase can lose activity above 60°C.
5. Test method transparency
Ask whether the supplier uses a fibrin plate assay, a validated in-house adaptation, or another unit system. Unit claims without method details are slippery.
6. Batch consistency
One shiny pilot lot proves very little. Commercial buyers need repeated lots with similar unit values, similar moisture, similar appearance, and stable handling behavior.
Different buyers, different purity priorities
This is where the conversation gets more practical.
A supplement brand may care most about activity-per-capsule, label story, and consistency. A nutraceutical wholesaler may care about storage stability and price per active unit. A pharma-oriented buyer will usually care about method traceability, impurity control, and whether performance claims survive deeper scrutiny.
Same ingredient. Different pressure points.
So the “right” purity level is not always the highest possible one. It is the purity level that supports your target use without hurting activity, stability, or cost structure.
For example:
- Dietary supplement brands often benefit from a balanced spec: strong activity units, sensible protein enrichment, low moisture, and clean documentation.
- Pharmaceutical ingredient buyers may push further on characterization, residuals, and process control—but still need proof that refinement has not weakened the active enzyme system.
- Traders and distributors should pay special attention to assay harmonization, because mismatched unit systems can wreck downstream customer trust.
You know what? That last one causes more headaches than people admit.
Red flags that purity is being used as a sales prop
A few warning signs show up again and again:
- Purity is highlighted, but activity units are buried
- The COA lists protein percentage but not specific activity
- The supplier cannot explain the assay method
- There is no mention of dry basis
- Processing sounds harsh, but no stability data is offered
- Marketing says “high potency,” while technical documents stay vague
- Batch-to-batch variance is hand-waved away
If you see three of those in one supplier file, slow down.
The smarter sourcing mindset
The best buyers don’t ask, “What’s your purity?”
They ask, “What purity profile gives me the most reliable activity in my application?”
That sounds like a small wording shift. It isn’t.
It moves the conversation from appearance to function, from broad protein claims to measurable fibrinolytic effect, and from price-per-kilo to value-per-active-unit.
That’s how serious sourcing teams avoid false economy.
Because in lumbrokinase, performance is not the same as cleanliness. The ingredient has to stay enzymatically alive, analytically credible, and commercially workable. If purity supports that, great. If purity comes at the expense of activity, then it’s the wrong kind of purity.
And that, really, is the whole game.
FAQs
1. Does higher purity always mean stronger lumbrokinase?
No. Higher purity only helps when it preserves and enriches the active fibrinolytic enzyme fraction. For sourcing, lumbrokinase activity units and specific activity are usually more useful than a purity claim by itself.
2. What activity units should buyers check when sourcing lumbrokinase?
Your uploaded testing method states a dry-basis requirement of not less than 12,000 units per mg, plus specific activity not less than 20,000 units per mg protein. Those are strong reference points for evaluating a lumbrokinase supplier.
3. Why is specific activity important in a lumbrokinase COA?
Because specific activity of lumbrokinase shows how much fibrinolytic performance is delivered relative to protein content. It helps distinguish a genuinely potent enzyme fraction from a powder that is simply high in total protein.
4. Can processing reduce lumbrokinase performance even if purity is high?
Yes. Your materials indicate that lumbrokinase can lose activity above 60°C, so aggressive heat exposure or drying can reduce enzyme performance even when the material appears cleaner or more refined.
5. What is the best way to compare lumbrokinase suppliers?
Compare suppliers using the same fibrin-based activity method, then review units per mg, specific activity, moisture, dry-basis reporting, and batch consistency together. That gives a more reliable picture than purity percentage alone.
