Before anyone talks about branding, pack sizes, or distributor margins, there’s a more basic question: can your earthworm protein powder legally sit on the shelf in the market you want to enter? That sounds obvious, sure. But with earthworm-derived ingredients, the answer changes fast depending on whether the product is positioned as a conventional food, a dietary supplement, a novel food, a pharmaceutical raw material, or even a cosmetic active.
And that’s the whole story, really. Earthworm protein powder is scientifically interesting and commercially intriguing, but globally, it is not a “one dossier fits all” ingredient.
Earthworm proteins are being studied as a high-protein raw material and as a source of bioactive peptides with antioxidant, ACE-inhibitory, and immunomodulatory activity. In the uploaded research, dried earthworm material showed protein levels around 60% to 70% depending on the study, and hydrolyzed fractions yielded small peptides linked to antioxidant, antihypertensive, and immune-related effects.
That scientific promise helps the business case. It also creates regulatory friction. Why? Because the stronger the bioactivity story sounds, the more likely regulators are to ask whether you are selling food, supplement, or something edging toward a drug claim. And that line matters a lot.


Start with the category, not the ingredient
Here’s the thing: earthworm protein powder may be one material, but regulators won’t see it as one material everywhere.
In the United States, FDA regulates dietary supplements under a framework different from conventional foods and drugs. A supplement is meant to supplement the diet, while a product intended to treat, diagnose, cure, or prevent disease is treated as a drug. FDA also notes that manufacturers or distributors of a new dietary ingredient not present in the food supply as an article used for food must submit a premarket safety notification before marketing. Dietary supplement CGMPs under 21 CFR Part 111 also apply to manufacturing, packaging, labeling, and holding. (U.S. Food and Drug Administration)
So if a brand wants to sell earthworm protein powder in capsules in the U.S., the first compliance question is not “Does the powder test high in protein?” It is “What is the legal ingredient status, and does it trigger a New Dietary Ingredient pathway?”
In the European Union, the first gate is often novel food status. The European Commission defines novel food broadly as food not consumed to a significant degree by humans in the EU before 15 May 1997, and the current novel food framework sits under Regulation (EU) 2015/2283. The Novel Food Catalogue is a non-binding but important screening tool, and the Union list contains authorised novel foods with their use conditions and specifications. (Food Safety)
Australia and New Zealand follow a similar logic. FSANZ says non-traditional foods that require safety assessment before being added to the food supply are novel foods, and anyone wanting to sell a novel food ingredient must apply for the Code to be amended. (foodstandards.gov.au)
Singapore also uses a pre-market approval framework for novel foods. SFA states that a novel food must be covered by pre-market approval before it can be lawfully supplied, handled for supply, or reared or cultivated there. SFA also invites companies to check whether a product is considered novel before filing. (Default)
That’s why global strategy begins with classification. Not marketing language. Not formulation. Classification.
Species identity is not a small detail
Honestly, this part gets underestimated all the time.
For earthworm-derived ingredients, species identity is not just a technical note in a COA. It can shape safety review, historical-use arguments, pharmacopoeial references, and customer confidence. One uploaded study identified the research material as Pheretima aspergillum, while another review notes that several Pheretima species are recognized in the Chinese Pharmacopeia for medicinal use.
That means suppliers should not casually blur “earthworm protein” into a generic commodity term. Buyers will want to know:
- exact species
- cultivation method
- feed or substrate controls
- harvest and cleaning procedures
- whether the ingredient is whole-earthworm powder, enriched protein, hydrolysate, or peptide fraction
And yes, those differences matter. Your own production-flow file describes a process involving selection, mechanical separation from soil and debris, secondary cleaning, washing/hydrolysis, centrifugation and filtration, low-temperature drying, milling, sterilization, and packing. It also lists added materials such as corn starch and maltodextrin.
That last point is a quiet but important one. If the commercial powder contains carriers, the regulatory review is no longer just about the earthworm protein. It becomes a finished ingredient-specification question. Purity, standardization, excipient disclosure, and label identity all get more complicated.
Safety dossiers win markets, not just protein percentages
You know what? A high protein number looks great in a brochure. Regulators care more about risk control.
The uploaded research is useful here because it shows that earthworm proteins can be processed into more defined fractions: purified protein, gastrointestinal digests, autolysates, and peptide-rich fractions. One study reported purified earthworm protein at 96.03% protein after alkali-soluble acid precipitation, while another showed that simulated digestion increased the share of low-molecular-weight material below 1 kDa from 44.80% to 80.19%.
That’s scientifically interesting. It also tells regulators that manufacturing conditions materially change the ingredient profile. So a credible safety package usually needs more than one test report.
For global markets, a serious dossier often needs to cover:
- identity and specification
- manufacturing process and process controls
- microbiological safety
- heavy metals and environmental contaminants
- pesticide or veterinary-drug relevance, if any
- allergen risk assessment
- toxicology or tolerated intake data where needed
- stability under intended storage and transport conditions
- intended use level and target population
And because earthworms live in close contact with soil and organic substrates, contaminant control becomes especially important. The 2024 review of earthworm extract even lists inorganic elements such as lead, cadmium, mercury, nickel, barium, and manganese among the chemical composition concerns discussed in the literature.
That does not mean earthworm protein powder is unsafe. It means the source environment and purification process must be documented tightly. No shortcuts. No hand-waving.
Claims are where many launches go sideways
This is probably the most commercial point in the whole article.
Research on earthworm-derived materials points to antithrombotic, antioxidant, antihypertensive, antimicrobial, wound-healing, and immunomodulatory potential. The review literature is broad, and the uploaded papers add specific peptide data for ACE inhibition, antioxidant activity, and immune-related activity.
But that is exactly why claim strategy needs discipline.
In the U.S., FDA allows structure/function claims for dietary supplements, but disease claims push products toward drug territory. Firms using certain structure/function claims must notify FDA within 30 days after first marketing the product with the claim. (U.S. Food and Drug Administration)
In the EU, health claims are tightly controlled, and the European Commission’s Register is the reference point for authorised and non-authorised claims. Foods and supplements cannot simply borrow pharmacology language from academic papers and print it on pack. (Food Safety)
So phrases like “dissolves blood clots,” “treats hypertension,” or “prevents thrombosis” may sound persuasive in a sales sheet, but they can trigger a drug-style compliance problem very quickly. For B2B suppliers, this is where many otherwise promising projects get stuck. The science sounds exciting; the wording gets reckless.
A smarter route is to separate:
- internal scientific substantiation,
- customer technical documents,
- finished-product marketing claims.
Those are not the same thing, and they shouldn’t be written the same way either.
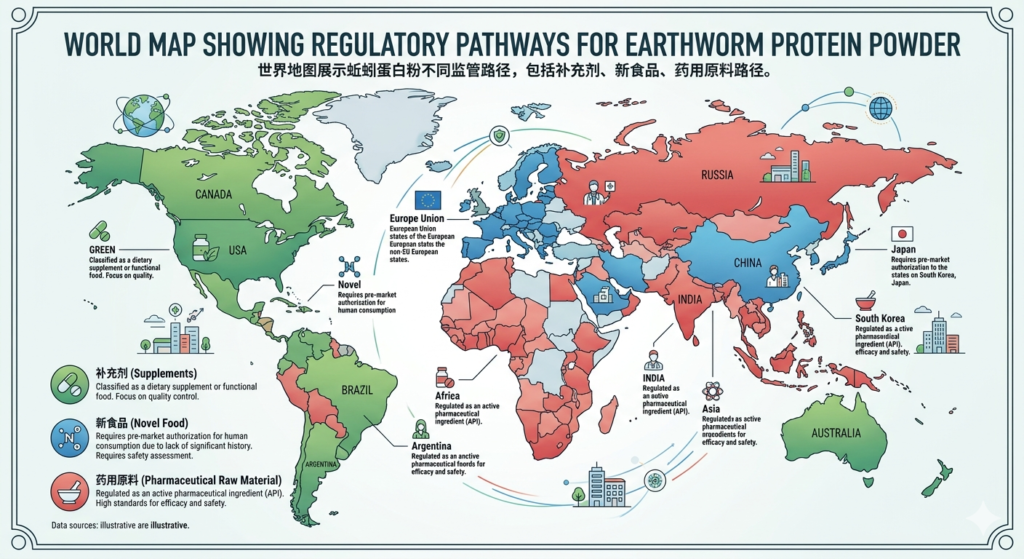
A practical market-by-market view
United States: possible, but only with careful ingredient-status work
The U.S. can be attractive because supplements have an established route. But earthworm protein powder is not a casual entry ingredient. FDA’s framework means you need to establish lawful ingredient status, assess whether an NDI notification is needed, manufacture under Part 111 CGMPs, and keep claims away from disease treatment language. (U.S. Food and Drug Administration)
For many suppliers, this is workable, but only after a real regulatory review.
European Union: likely the toughest food route
If significant EU consumption before 15 May 1997 cannot be shown, novel food review becomes the central issue. And even after market access, claims remain tightly restricted through the EU health-claims system. (Food Safety)
In plain English: the EU is promising, but not a quick launch market.
Singapore: innovation-friendly, but dossier-heavy
Singapore is open to novel food innovation, yet the route is explicitly pre-market and evidence-based. That makes it a good test market for serious companies with strong safety files, not for speculative exporters with thin paperwork. (Default)
Australia and New Zealand: similar story, safety first
FSANZ’s novel food approach means the product needs to clear a pre-market safety lens before broader food use. For suppliers, that usually means planning early and budgeting for data. (foodstandards.gov.au)
China and parts of East Asia: more familiarity, but not automatic freedom
The uploaded research notes that earthworm protein has been recognized as a new food resource in China since 2009, and the broader review shows long-standing medicinal use in East Asia.
That said, suppliers still need to match the intended commercial route to the right category: food ingredient, health food material, pharmaceutical input, or cosmetic raw material. History helps, yes. It does not replace dossier discipline.
What serious buyers should ask suppliers
If you’re a supplement brand, ingredient trader, or private-label manufacturer, don’t stop at “Do you have a protein test report?”
Ask for the full compliance picture:
- species name and origin
- breeding and substrate records
- manufacturing flowchart
- full specification sheet
- contaminant panel
- micro limits
- batch traceability
- carrier or excipient disclosure
- claim-support boundaries
- target-market regulatory assessment
That may sound like a lot. It is. But global buyers are not really buying powder. They’re buying risk-managed market access.
And with earthworm protein powder, that’s the real product.
So, is earthworm protein powder suitable for global markets?
Yes, but with a big asterisk.
It is suitable where the company treats regulation as part of product development, not as paperwork after the sale. The ingredient has a plausible commercial future because it combines high protein content with a growing body of research around bioactive fractions and functional-food potential.
Still, global suitability won’t come from enthusiasm alone. It will come from species clarity, controlled production, strong safety files, careful claims, and market-by-market category planning.
A bit unglamorous? Maybe. But that’s how real ingredient businesses scale without getting burned.
FAQs
1. Is earthworm protein powder considered a novel food in many countries?
In many markets, yes, it may be treated as a novel or non-traditional food ingredient if there is not enough documented history of safe human consumption in that jurisdiction. The EU, Singapore, and Australia/New Zealand all use pre-market safety review mechanisms for novel foods. (Food Safety)
2. Can earthworm protein powder be sold as a dietary supplement in the U.S.?
Potentially, but U.S. sellers still need to confirm lawful ingredient status, assess whether a New Dietary Ingredient notification is required, follow dietary supplement CGMPs, and avoid disease-treatment claims on label and marketing. (U.S. Food and Drug Administration)
3. What safety tests are most important for imported earthworm protein powder?
The big ones are identity, microbiology, heavy metals, environmental contaminants, process controls, and stability. For earthworm-derived ingredients, source-environment control is especially important because the raw material is closely tied to soil and substrate conditions.
4. Are cardiovascular or thrombosis claims allowed for earthworm protein ingredients?
Usually not as straightforward marketing claims for foods or supplements. Academic studies discuss ACE inhibition, fibrinolytic activity, and antithrombotic mechanisms, but those kinds of statements can push a product toward drug-style regulation if used carelessly in commerce. (U.S. Food and Drug Administration)
5. What should buyers ask before sourcing earthworm protein powder for global distribution?
Ask for species identity, cultivation records, processing flow, carrier disclosure, contaminant testing, batch traceability, market-specific regulatory assessment, and guidance on compliant claim language. For global launches, those details matter as much as price and protein percentage.
