Quick outline
- What makes lumbrokinase tricky, and valuable, in formulation
- Raw material form, enzyme activity, and why process control matters
- Dosage form choices: capsules, tablets, sachets, and blends
- Protecting enzyme performance during manufacturing and storage
- Compatibility with excipients and partner actives
- Labeling, positioning, and technical questions B2B buyers should ask
- Final takeaways for supplement, nutraceutical, and pharma-focused teams
- FAQs

Lumbrokinase sounds simple on paper. It is usually described as a fibrinolytic enzyme complex derived from earthworm sources, valued for thrombolytic and circulation-related applications. But once you move from concept to real product development, things get a bit more demanding.
That’s where formulation starts to matter. A lot.
Lumbrokinase is not the kind of ingredient you toss into a formula and hope for the best. It is enzyme-based, activity-sensitive, and closely tied to how you process it, protect it, combine it, and present it. Research and reviews on earthworm-derived bioactives consistently point to lumbrokinase as a potent fibrinolytic component with strong antithrombotic relevance, and they also note the importance of oral delivery and ongoing work around clinical application.
So, for supplement brands, pharmaceutical ingredient buyers, and contract manufacturers, the real question is not just “Can we use lumbrokinase?” It is “How do we use it without wrecking the thing that makes it valuable?”
Let me explain.
The first thing to remember: lumbrokinase is an enzyme, not just a label claim
This sounds obvious, yet it changes everything.
Lumbrokinase belongs to a group of fibrinolytic enzymes isolated from earthworm sources. Reviews in the uploaded materials describe it as a powerful fibrinolytic enzyme, note its thrombolytic and anticoagulant relevance, and mention oral formulations as an important route of use.
That enzyme nature means your formulation work has to protect functional activity, not merely ingredient presence. In other words, a product can contain lumbrokinase on paper and still underperform if the enzyme loses activity during blending, compression, drying, transport, or shelf storage.
Honestly, that is where many ingredient discussions go sideways. Teams focus on milligram count, while buyers with experience start asking the sharper question: what happens to activity after processing?
That’s the real game.
Raw material quality comes before clever formulation

Before choosing capsule shells or talking about release profiles, you need to understand the starting material.
The files here show that earthworm-derived protein materials are produced through steps such as selection, cleaning, hydrolysis, centrifugal separation, low-temperature drying, milling, sterilization, and packaging. Even though that specific process file refers to earthworm protein powder more broadly, it highlights something highly relevant to lumbrokinase formulation too: gentle handling and thermal control are not side issues. They are central.
Why? Because enzymes are fussy. A little too much heat, moisture, shear, or poorly chosen excipient exposure, and performance can drop.
So when sourcing lumbrokinase, product developers should look at:
- enzyme activity specification, not just ingredient weight
- source standardization and batch consistency
- upstream drying and purification conditions
- residual moisture and flow characteristics
- microbial and heavy metal controls
- packaging that limits heat and humidity stress
You know what? If the raw material is unstable, no “smart” formula will magically rescue it.
Oral delivery is promising, but it shapes the formulation strategy
One especially useful point in the review literature is that lumbrokinase is discussed as an oral formulation ingredient, with intestinal absorption specifically mentioned in the cited review material. That matters because it gives formulators a practical frame: this is not only a lab enzyme. It has real oral product relevance.
Still, oral delivery is not one-size-fits-all.
Capsules usually make the cleanest starting point
For most B2B projects, capsules are the easiest and safest entry format for lumbrokinase.
Why capsules?
Because they reduce mechanical stress compared with hard-compressed tablets. Compression can be rough on delicate actives. With an enzyme complex, that should make you pause. Capsules also simplify early-stage pilot work, speed up line transfer, and make dose adjustment easier during commercial refinement.
That is why lumbrokinase often fits naturally into:
- hard capsules for targeted circulation support products
- capsule-in-carton medical-style nutraceutical formats
- combination enzyme formulas aimed at premium channels
There is also a practical market signal in the uploaded material referencing “lumbrokinase enteric capsules” in clinical use discussion. That does not mean every product must be enteric-coated, but it does show why delayed protection is a recurring formulation topic.
Tablets can work, but only with care
Can you make tablets? Sure. Should you rush into it? Probably not.
Tablets are attractive for cost, consumer familiarity, and shipping efficiency. But they bring more heat, pressure, and friction during production. For a sensitive enzyme ingredient, that can become a headache.
If a tablet route is necessary, formulators usually need to think harder about:
- compression force
- dwell time
- granulation method
- binder selection
- lubricant balance
- post-compression protection
A strong tablet that looks great but delivers weaker enzyme performance is not really a win.
Sachets and powder sticks are possible, but more niche
Sachets can work in some markets, especially when brands want premium clinical-style positioning or combination formulas. But enzyme taste, moisture exposure after opening, and compatibility with powdered partner ingredients can make this format trickier.
For most brands, sachets are a second-phase innovation, not the first launch choice.
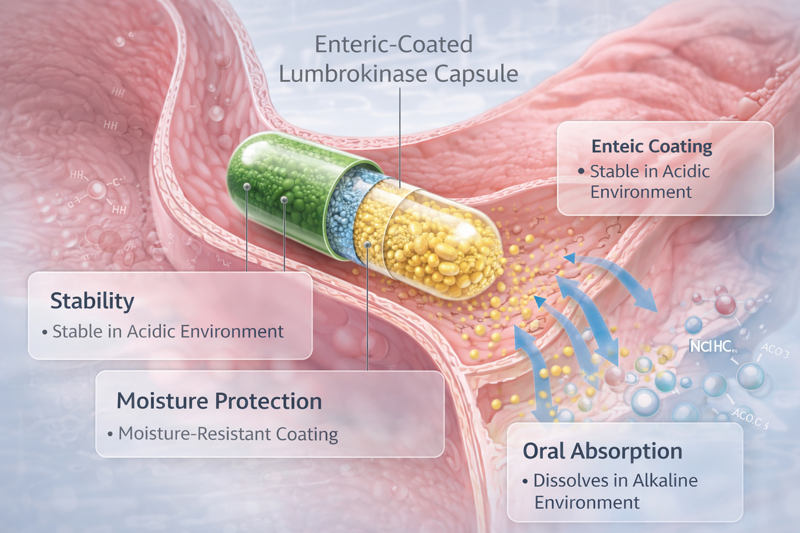
Enteric protection keeps coming up for a reason
Here’s the thing: when people talk about lumbrokinase formulation, enteric delivery keeps showing up because it addresses a practical challenge. Oral enzymes may need protection through the stomach environment before release further down the GI tract.
The uploaded content mentions lumbrokinase enteric capsules in applied use discussion, and the broader review also notes oral formulation relevance.
That does not automatically mean every market, every claim set, or every formulation must go enteric. But it does mean developers should seriously evaluate:
- whether gastric exposure may reduce desired activity
- whether delayed release improves product logic
- whether the cost increase is justified by positioning and performance goals
- whether the target market expects a more clinical delivery system
For premium supplement lines, enteric-coated capsules can strengthen both technical credibility and commercial story. For lower-cost channels, standard capsules may still be the better business choice.
A little contradiction here: simpler formats often sell better, yet more protective formats may perform better. That tension is normal. Formulation is often a balancing act between elegant science and workable business reality.
Heat, humidity, and process stress are the quiet troublemakers
If you only remember one manufacturing point, make it this: avoid unnecessary stress.
The earthworm protein process file specifically references low-temperature drying. That detail fits the broader logic of working with sensitive bioactive materials. Whether you are handling purified enzyme fractions or enzyme-rich earthworm-derived ingredients, the same practical lesson applies: high heat is rarely your friend.
Formulators should pay close attention to:
1. Temperature exposure
Drying, blending, coating, transport, and warehousing conditions all matter. Even brief heat spikes can reduce functional quality.
2. Moisture control
Enzymes and humidity are not a happy couple. Moisture can affect flow, stability, and activity. Use good desiccant strategy and barrier packaging where appropriate.
3. Mechanical stress
Aggressive milling, extended blending, or heavy compression may damage performance.
4. Hold time during production
The longer an unfinished blend sits in poor environmental conditions, the more risk builds.
This is not glamorous stuff. It is factory-floor stuff. But it is often what separates a reliable finished product from a disappointing one.
Excipients matter more than people think
A lumbrokinase formula is not just lumbrokinase. It is lumbrokinase plus every other decision you make.
Fillers, binders, lubricants, disintegrants, coating systems, flavors, sweeteners, anti-caking agents, and carrier powders can all affect stability or manufacturability. Some combinations behave beautifully. Some are a mess.
In early development, teams should screen for:
- moisture contribution from excipients
- pH-related compatibility concerns
- adsorption losses on carriers
- flow support without over-lubrication
- capsule fill uniformity
- coating materials that do not compromise activity
And yes, sometimes the most boring excipient shortlist is the smartest one. Fancy formulas are exciting in meetings. Stable formulas are the ones that survive scale-up.
Combination formulas need logic, not clutter
Lumbrokinase is often considered for circulation, vascular health, or broader cardio-support positioning. That makes combination formulas tempting.
But not every “heart health” add-on belongs in the same capsule.
When brands stack too many partner actives, a few problems show up fast:
- reduced room for meaningful enzyme loading
- more compatibility unknowns
- higher odor or taste burden
- more difficult stability work
- muddier product positioning
A cleaner approach is usually better. Pair lumbrokinase only with ingredients that support a clear product story and do not create avoidable formulation stress.
For pharmaceutical ingredient buyers or advanced nutraceutical teams, this is where pilot studies earn their keep. Compatibility work is not wasted time. It is insurance.
Regulatory and positioning questions should shape the formula early
A formula is never just technical. It is also regulatory and commercial.
The uploaded review frames lumbrokinase within antithrombotic and thrombolytic contexts, with ongoing clinical interest and oral formulation relevance. That is useful scientifically, but it also means brands need to be careful about how they position finished products in different markets.
So before locking the formula, ask:
- Is this a dietary supplement, a nutraceutical, or a pharma-adjacent product?
- What claims are realistic in the target country?
- Does the dosage form need to look more “clinical” for the sales channel?
- Will buyers expect enteric delivery, activity units, or both?
- Are you selling to e-commerce consumers, practitioners, or institutional buyers?
Same ingredient. Different formulation logic.
A practitioner-focused SKU may lean toward a clean enteric capsule with tighter technical storytelling. A retail supplement may need a simpler presentation and easier price point. A bulk ingredient sale to manufacturers may focus less on front-label marketing and more on activity retention through downstream processing.
What experienced buyers usually ask suppliers
B2B buyers tend to cut through the fluff pretty fast. They often want answers to questions like:
- What is the activity specification per gram or per serving?
- How stable is the ingredient under normal production conditions?
- Do you have data after encapsulation or tableting?
- Is enteric delivery recommended or simply optional?
- What packaging protects the ingredient best?
- Which excipients have already been screened?
- What are the storage conditions and expected shelf life?
Those are good questions. In fact, they are the questions.
If your supplier can answer them clearly, formulation gets easier. If not, you may be buying uncertainty dressed up as innovation.
So, what should brands keep front and center?
Here’s the simple version.
Lumbrokinase can be a compelling ingredient for circulation-focused and premium health products because it is associated with fibrinolytic activity, oral formulation interest, and long-standing research attention in earthworm-derived bioactives. But it is also an enzyme-sensitive material, which means formulation choices directly affect commercial value.
That leads to a few practical conclusions:
Capsules are often the best starting dosage form.
Enteric protection deserves serious consideration.
Low-stress processing is not optional.
Excipient compatibility needs real work.
Activity retention matters more than label weight.
And raw material quality sets the tone for everything that follows.
Honestly, that is the heart of it.
A lumbrokinase product can look premium, sound scientific, and fit a strong market trend. But if the formulation does not respect the ingredient, the product may never reach its real potential.
Related reading idea: How to Evaluate the Quality of Lumbrokinase
Related reading idea: How Brands Integrate Lumbrokinase into Existing Product Lines
FAQs
1. What is the most suitable dosage form for lumbrokinase supplements?
For many brands, capsules are the most practical dosage form for lumbrokinase supplements because they place less mechanical stress on enzyme ingredients and are easier to adapt during pilot and commercial production.
2. Does lumbrokinase need enteric coating?
Not always, but enteric coating is often considered for oral lumbrokinase formulations because it may help protect sensitive enzyme activity before release further along the digestive tract.
3. Why is enzyme activity more important than ingredient weight in a lumbrokinase formula?
Because lumbrokinase is an enzyme complex, performance depends on retained functional activity, not just how many milligrams appear on the label. That is why activity testing and post-processing stability matter so much.
4. What manufacturing conditions can reduce lumbrokinase stability?
High heat, excess humidity, aggressive compression, long hold times, and poorly matched excipients can all reduce lumbrokinase stability during production and storage.
5. What should B2B buyers ask before sourcing lumbrokinase raw material?
Buyers should ask about activity specification, batch consistency, recommended dosage forms, excipient compatibility, storage conditions, packaging protection, and whether stability data exists after encapsulation or tableting.
