Quick article skeleton
- Why lumbrokinase keeps showing up in B2B product planning
- What makes it commercially interesting beyond the science
- Four case-study-style B2B application scenarios
- What buyers usually need from a supplier before moving ahead
- Where the market is likely heading next
- FAQ

Case Study: Successful B2B Applications of Lumbrokinase
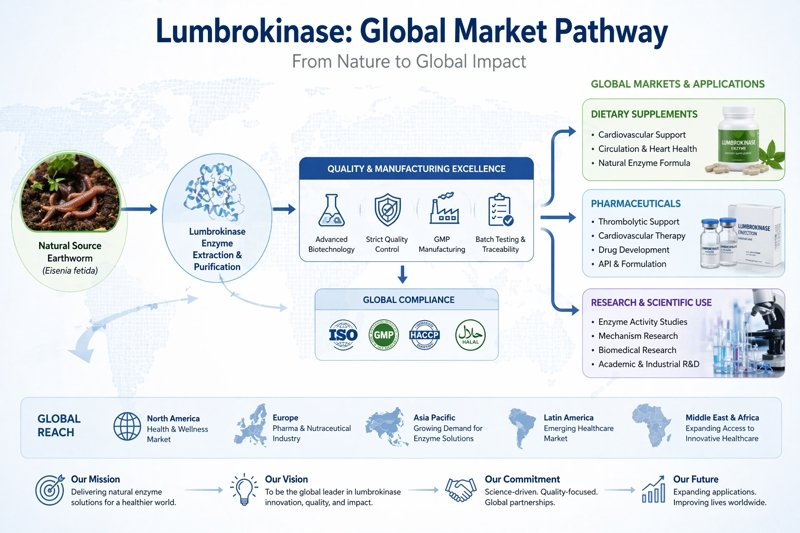
Lumbrokinase has moved far beyond being a niche enzyme that only a handful of formulators talked about. For B2B buyers in dietary supplements, nutraceutical ingredients, pharmaceutical intermediates, and advanced health formulations, it has become one of those ingredients that sparks a very practical question: can this enzyme actually fit a real product pipeline, a real sourcing plan, and a real compliance review?
That is where things get interesting.
Scientifically, lumbrokinase is widely described as an earthworm-derived fibrinolytic enzyme complex with a dual mechanism. It can act as a plasminogen activator and also participate in direct fibrin degradation, which is a big part of why it keeps attracting attention in circulation-focused formulation work. The material in our product file positions it the same way, highlighting high fibrinolytic activity, targeted enzymatic action, and value for nutraceutical, pharmaceutical, and functional health product development. Our broader review article also notes that lumbrokinase has long been studied for antithrombotic and thrombolytic potential, and discusses its reported advantage of acting with less tendency toward hyperfibrinolysis-related bleeding than less selective fibrinolytic approaches.
For B2B buyers, though, the story is never just mechanism. It is supply chain, assay clarity, formulation stability, regulatory positioning, label strategy, and whether the ingredient can survive the messy middle between a promising concept and a finished SKU.
So let’s look at successful B2B applications of lumbrokinase through a case-study lens. Not with invented brand names or glossy fairy tales, but with realistic application models grounded in the product and research materials you provided.
Why buyers keep circling back to lumbrokinase
Here’s the thing: lots of ingredients sound exciting in a lab report. Fewer survive commercial scrutiny.
Lumbrokinase keeps showing up because it sits at the intersection of three forces buyers care about:
- It has a clear functional story tied to fibrin-related activity and circulation-oriented product concepts.
- It fits the continuing market interest in enzyme-based and bioactive ingredients derived from natural sources.
- It gives manufacturers room to build differentiated specifications, especially around activity ranges, formulation type, and end-use positioning. Our product’s activity specifications from 10,000 IU/mg to 200,000 IU/mg, which already hints at how important spec matching is in B2B negotiations.
And honestly, that flexibility matters. Some buyers are not looking for the “highest possible activity.” They are looking for the right activity, with the right delivery format, at the right cost, with the right paperwork.
That is a very different conversation.
Case study 1: A dietary supplement brand building a circulation-support capsule
This is probably the cleanest B2B use case.
A supplement brand wants to launch a premium circulation-support product. The marketing team wants something more distinctive than the usual vitamin-plus-herb formula. The formulation team wants an active ingredient with a known mechanism story. Procurement wants batch consistency. Regulatory wants claims kept in check.
Lumbrokinase fits because it gives the brand a clear enzyme-centered identity. The positioning of our Lumbrokinase products, the ingredient is explicitly positioned for cardiovascular support capsules, circulation formulas, and enzyme complexes.
Why this application works
The appeal here is not just that lumbrokinase sounds advanced. It is that it helps a brand create a more technical, more premium product narrative. A capsule built around enzyme activity feels different from a generic circulation blend. That difference can matter a lot in distributor conversations and private-label presentations.
The successful B2B move in this kind of case usually depends on four decisions:
Activity standard selection
Buyers need a spec that matches their formulation goal. A premium line may want higher activity material, while a broader retail product may choose a more moderate standard to control cost and capsule fill weight. Your product file’s wide activity range shows why this step is central, not optional.
Assay transparency
This is where deals can slow down. Enzyme ingredients live or die by testing clarity. Buyers want to know the assay method, the unit system used, and whether the COA reflects real production variability or a best-case number. In our previous articles, we have also touched upon the procurement pain points within the industry: unclear assay standards.
Low-temperature process control
The same file emphasizes controlled processing, including low-temperature drying, as essential for maintaining enzyme stability. That matters because enzyme buyers are usually a bit skeptical by default — and not without reason.
Shipping confidence
Again, very practical. Your product sheet calls out activity loss after shipping as a hidden sourcing risk and positions proper packaging and storage as part of the answer.
What success looks like
In a successful supplement application, lumbrokinase is not sold as a miracle. It is sold as a premium technical ingredient supported by stable activity, documentation, and a clear capsule-ready positioning. The buyer wins because the finished product looks more specialized. The supplier wins because the ingredient is harder to replace with a commodity alternative.
That is good chemistry, in every sense.
Case study 2: A contract manufacturer creating a multi-ingredient enzyme complex
Now we get into a more nuanced use case.
Some contract manufacturers are not building a single-ingredient flagship. They are helping brand clients develop complex formulas — sometimes for circulation, sometimes for “healthy aging” or broader wellness positioning. In these formulas, lumbrokinase may be one part of a stacked system rather than the whole headline.
Our lumbrokinase product documentation indicates directly notes compatibility with enzyme blends and broader functional products, including anti-aging formulations and blood-flow support products.
Why this application works
From a formulation standpoint, lumbrokinase brings a focused technical role into a broader formula architecture. That can be useful when the brand wants something smarter than a one-note product.
But this model only works when the supplier and contract manufacturer are aligned on process details.
Because enzyme blends can be fussy. Very fussy.
Heat, moisture, compression force, excipient choice, and packaging all start to matter more than people expect. Your file’s emphasis on controlled extraction, filtration, low-temperature drying, sterilization, and packaging reflects this reality.
Where the B2B value shows up
For contract manufacturers, successful use of lumbrokinase in blends usually depends on:
- stable incoming activity
- predictable behavior during blending and encapsulation
- a supplier that can explain not just the ingredient, but how it behaves in real manufacturing
- spec customization when a customer needs a certain activity range rather than a one-size-fits-all option. Your uploaded product material explicitly mentions customizable specifications and formulation support.
This last point is bigger than it sounds. A rigid supplier creates friction. A technical supplier with some flexibility becomes part of the customer’s development workflow.
That is often the difference between a quote request and a long-term account.
Case study 3: A pharmaceutical or research buyer sourcing lumbrokinase for thrombolytic research
This is a more specialized channel, but an important one.
Lumbrokinase has been studied as a potent fibrinolytic system and mentions that oral lumbrokinase formulations have reached clinical-trial approval stages in several countries. Your lumbrokinase product page also positions the ingredient for thrombolytic research and circulatory health studies.
That combination matters because research buyers do not purchase like supplement brands do. They are usually more exacting about source control, bioactivity documentation, and purity-related process discussion.
Why this application works
Research buyers are not impressed by marketing gloss. They want technical credibility.
What helps lumbrokinase here is that the scientific literature around earthworm-derived bioactives is broader than one single mechanism. The review summarizes not just antithrombotic activity, but a wider family of bioactive functions found in earthworm extracts, including antioxidant, wound-healing, and antifibrotic directions. That broader scientific environment makes lumbrokinase more interesting as a platform ingredient, not just a one-off raw material.
What research buyers usually ask for
They tend to care about:
- raw material origin
- process consistency
- activity testing method
- storage conditions
- batch reproducibility
- microbial and heavy-metal controls
Our product includes many of these decision points already, from source material and pH range to moisture, ash, selenium, arsenic, cadmium, lead, mercury, and microbiological limits.
So in this application, success is not just “the buyer purchased.” Success is that the material is documented well enough to enter a formal R&D workflow without constant back-and-forth.
That saves time. And in technical buying, time saved is often the hidden sale.
Case study 4: A distributor building a differentiated enzyme portfolio
Distributors look at lumbrokinase a bit differently.
They are not always formulating it themselves. They are asking whether it belongs in a portfolio that serves supplement factories, lab buyers, and regional brands. In that world, the value of lumbrokinase lies in differentiation. It is not another generic vitamin. It is not another crowded botanical. It is an enzyme with a specialized function story and stronger technical barriers.
Why this application works
Distributors need ingredients that help them sound credible in front of customers. Lumbrokinase does that because it immediately opens a deeper conversation:
- What activity do you need?
- What assay method are you comparing?
- Capsule or tablet?
- Domestic production or export?
- Standard spec or custom requirement?
That kind of conversation creates stickier B2B relationships than commodity ingredients usually do.
Our lumbrokinase supports this positioning by focusing on scalable supply, batch verification, customizable specifications, and technical support.
What makes the application successful
A distributor succeeds with lumbrokinase when they do not treat it like a simple catalog line. The best distributors build a mini-knowledge base around it:
- application notes
- typical activity ranges
- storage guidance
- packaging options
- sample policy
- lead-time clarity
- target customer segments
In other words, they sell confidence, not just powder.
And yes, that sounds obvious. But plenty of distributors still try to move technical ingredients with almost no technical selling. It rarely ends well.

The overlooked commercial lesson: lumbrokinase is a documentation-driven sale
You know what? This is probably the most important point in the whole article.
Lumbrokinase does not usually fail in B2B because the science is uninteresting. It fails when documentation is thin, assay language is vague, or the supplier cannot translate technical details into commercial reassurance.
That is why our own product file is right to stress hidden sourcing risks such as activity loss, batch inconsistency, unclear standards, and delayed delivery. Those are not side issues. They are the sale.
A successful B2B application of lumbrokinase usually rests on five practical pillars:
What buyers want before they commit
1. Clear activity language
They need to know what the number means, how it is tested, and how it compares across suppliers.
2. Process credibility
Low-temperature drying, filtration, controlled extraction, and packaging are not just manufacturing details. They are proof points.
3. End-use guidance
Capsules, enzyme complexes, research use, circulation-focused formulas — each route asks different questions.
4. Batch consistency
Without repeatability, even a brilliant first sample goes nowhere.
5. Supply realism
Stable bulk supply, export readiness, and practical lead times often decide who gets the order.
That is why successful lumbrokinase suppliers tend to behave more like technical partners than mere traders.
So where is the market heading?
The future of successful B2B lumbrokinase applications will likely be shaped by specialization, not mass-market sameness.
The research supports continued interest in earthworm-derived proteins and bioactive fractions as functional ingredients, including antihypertensive peptides, antioxidant peptides, and immunomodulatory hydrolysates. That wider ecosystem helps lumbrokinase, because buyers are increasingly looking at ingredient families, not isolated raw materials.
In practice, that means a few things are likely:
- more premium circulation-focused supplement formulas
- more technical scrutiny around assay and stability
- more demand for customized specs rather than standard catalog grades
- more supplier evaluation based on process control and support, not just price
A bit of a contradiction shows up here. As the ingredient becomes more commercially attractive, buyers also become more cautious. But that is not a bad sign. It usually means the market is maturing.
And mature markets reward suppliers who can explain, document, and deliver.
Final takeaway
The most successful B2B applications of lumbrokinase are not flashy. They are disciplined.
A supplement brand uses it to create a sharper circulation-support SKU.
A contract manufacturer uses it to strengthen an advanced enzyme blend.
A research buyer uses it because the mechanism and documentation justify investigation.
A distributor uses it to build a more technical, more defensible portfolio.
Different paths, same lesson: lumbrokinase works best commercially when science, specification, and supply move together.
That is the real case study.
FAQs
1. What are the most common B2B applications of lumbrokinase?
The most common B2B applications of lumbrokinase include circulation-support dietary supplements, enzyme complex formulations, thrombolytic research materials, and specialty health product development. Your uploaded product material also positions it for cardiovascular support capsules and circulatory health studies.
2. Why do supplement brands choose lumbrokinase over more common circulation ingredients?
Many brands choose lumbrokinase because it offers a more technical enzyme-based positioning, with a mechanism centered on fibrin-related activity rather than a generic wellness story. That helps create a differentiated premium supplement concept.
3. What should buyers check when sourcing bulk lumbrokinase powder?
Buyers should review activity specification, assay method, batch consistency, microbial and heavy-metal controls, storage requirements, and packaging conditions. These are critical when evaluating bulk lumbrokinase powder for supplements or research applications.
4. Is lumbrokinase suitable for custom specification projects?
Yes. For many B2B buyers, custom activity and formulation requirements are part of the sourcing process. Your uploaded lumbrokinase product file specifically notes customizable specifications and technical support for formulation.
5. What makes a lumbrokinase supplier more reliable for long-term B2B cooperation?
A reliable lumbrokinase supplier usually offers stable raw material sourcing, controlled processing, verified activity per batch, clear documentation, and dependable delivery planning. In long-term B2B cooperation, those points matter as much as price.

Get High-Activity Lumbrokinase For Your Formulations
Contact us immediately to discuss your project requirements.
